Mineral Flame Protection
|
General
|
 Products based on aluminium hydrate and aluminium fire disaster and consequently they save human lives. ATH
Aluminium Hydroxide
(Aluminium Trihydrate)
MDH
Magnesium Hydroxide
(Magnesium Dehydrate)
AOH
Aluminium Oxide-Hydroxide
(Aluminium Monohydrate)
|
| |
| Plastic products have become an integral part of everyday life, but due to their composition and structure most plastics are highly combustible. In order to come to grips with this problem, manufacturers are now adding flame retardants to more and more materials. These additives prevent or delay the development of fire or a flashover by heat with drawal, water release or by the formation of a barrier layer that keeps away the oxygen required for combustion. |
 |
| Another positive effect is that they reduce the flue gases arising from fires allowing more time to flee from the flames. Flame retardant plastics can prevent fire disaster with disastrous consequences caused by a occurrences such as a short circuit in electric power lines. A group of flame retardants are of mineral origin. They are based on metal hydrates, particularly on aluminium hydroxide and magnesium hydroxide. |
| |
 |
| |
|
Over the years they have become well established as the most important fire retardants due to their environmental friendliness (they are halogen-free) and their favourable price: performance ratio.
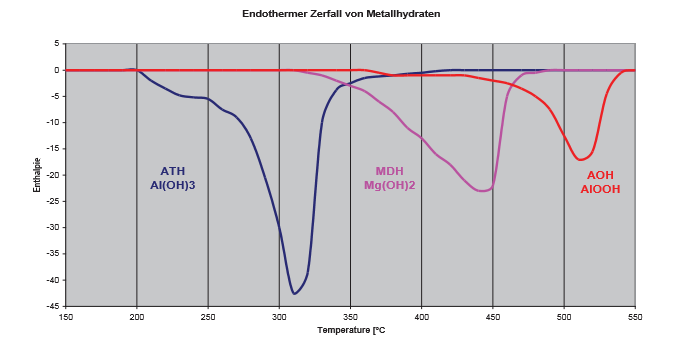
The flame-protecting effect of metal hydrates is based on a physio-chemical mechanisms. The diagram on page 28 shows the endothermic decomposition of metal hydrates in the case of fire. The decomposition of aluminium oxide is as follows: in the presence of an ignition source - a flame or a hot object - the thermal decomposition of the aluminium hydroxide into aluminium oxide and water takes place. During this process, energy is detracted from the ignition source, as the decomposition is an endothermic reaction.
At the same time, the released water vapour is cooling the surface of the polymer and it particularly dilutes the concentration of burnable gases in the surrounding area of the burning plastic.
The remaining aluminium oxide residue has a high internal surface where sooty particles, respectively polycyclic aromatic carbon hydrates (PAC) are absorbed. Additionally the oxide residue acts as a barrier, disabling the further release of low molecular weight decomposition products as well as a heat barrier protecting the polymer against further decomposition.
|
| |
Processing Temperature:
- ATH bis 200°C
- MDH bis 300°C
- AOH bis 320°C
|
Decomposition reaction of the metal hydrates
Aluminiumhydroxid
2 AI(OH)3 + 1075 kJ/kg ---> AI2O3 + 3 H2O
Aluminiumoxidhydroxid
2 AIOOH + 700 kJ/kg ---> AI2O3 + H2O
Magnesiumhydroxid
Mg(OH)2 + 1220 kJ/kg ---> MgO + H2O |
| |
| The decomposition reaction of the metal hydroxides starts at different temperatures. Aluminium hydroxide has the lowest decomposition temperature - the endothermic phase transition starts at around 200°C. Therefore, aluminium hydroxide is used only up to 200°C processing temperature. For higher process temperatures magnesium hydroxide and aluminium oxide are used. For a constant flame protection the lower energy requirement for the decomposition of aluminium hydroxide must be compensated with a higher degree of filling. This is willingly accepted as very good processing and final properties are obtained in combination with other flame retardants. |
| |
Bauxite - the most important aluminium ore
|
 |
| |
| Bauxite is the world's main source of aluminium. It consists of several aluminium minerals mainly of gibbsite (gibbsite, Al (OH) 3) and it is a transformation product of aluminium-rich rocks. In 1821 Bauxite was discovered by the French geologist Pierre Berthier in Southern France. He named the mineral after the place of discovery, the village Les Baux, Provence. Usually bauxite is strip mined because it is found near the surface. The Bayer process is the principal industrial process of bauxite refining. To produce aluminium (aluminium oxide) bauxite is digested by washing with a hot solution of caustic soda under pressure. The aluminium oxide of the ore is converted into soluble sodium aluminate and it is separated from insoluble impurities. The solution is cooled and high-purity aluminium hydroxide with a particle size of 100µ precipitates. This is the raw material for further processing. Mainly (from about 95 percent) metallic aluminium is produced. The rest is used in non-metallic applications. |